Legal Basis
- Pharmaceutical Law 2016 and Amended Pharmaceutical Law 2024.
- Price Law 2023.
- Decree 85/2024/ND-CP detailing certain provisions of the Price Law.
- Essential Medicines List: Circular No. 28/2024/TT-BYT.
- Non-Prescription Medicines List: Circular No. 07/2017/TT-BYT.
- Prescription Medicines List: Includes all medicines not listed in the non-prescription medicines list.
Drug Price Management Measures
– Detailed regulations in Article 107 of the Pharmaceutical Law and its amendments.
– Key differences compared to the 2016 Pharmaceutical Law:
- The 2016 Pharmaceutical Law only required price declaration and re-declaration.
- Amended Law 2024:
- Price management through wholesale price announcement and re-announcement.
- Price management through wholesale price declaration, retail price declaration, and re-declaration.
Forms of Drug Price Announcement and Declaration
1. Wholesale Price Announcement
- Announcing Entities:
- Importers (for foreign medicines),
- Manufacturers (for domestically produced medicines).
- Regulatory Authority: Ministry of Health.
- Timing of Announcement:
- Must be announced before selling the first batch to the market.
- Required Documents: Pending guidelines.
- Management Approach:
- Announced prices will be publicly available on the Drug Administration of Vietnam’s electronic portal.
- Announced prices represent wholesale prices for the entire distribution chain. No wholesaler is allowed to sell above the announced price.
- Estimated fee: 800,000 VND per submission.
- Effective Date: From July 1, 2025, except for previously declared and re-declared prices under the old law.
2. Wholesale and Retail Price Declaration
- Declaring Entities:
- Wholesale establishments,
- Retail establishments (pharmacies, drug counters, healthcare station medicine cabinets).
- Notes:
- Not all wholesale and retail establishments are required to declare prices.
- The list of required declaring entities will be issued periodically by the Provincial People’s Committee before February 15, 2024.
- Regulatory Authority: Submission to the Department of Health.
- Timing of Declaration:
- Within 5 days from the price determination date (invoice issuance).
- Management Approach:
- Prices will not be publicly disclosed. Instead, the Department of Health will compile them into the national price management database. Only authorized entities with accounts can access the data.
- Sales must follow the declared prices.
- No submission fees required.
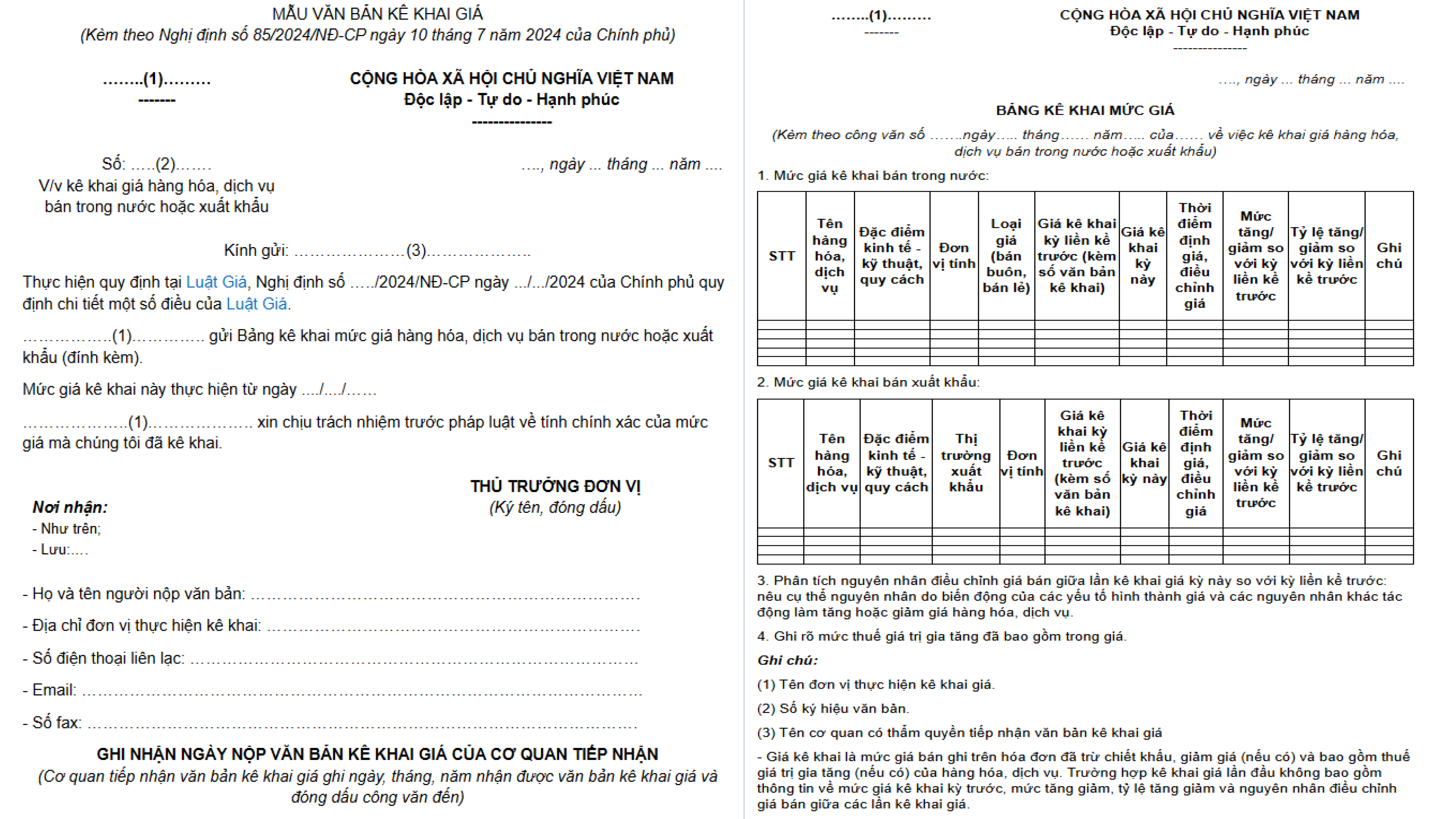
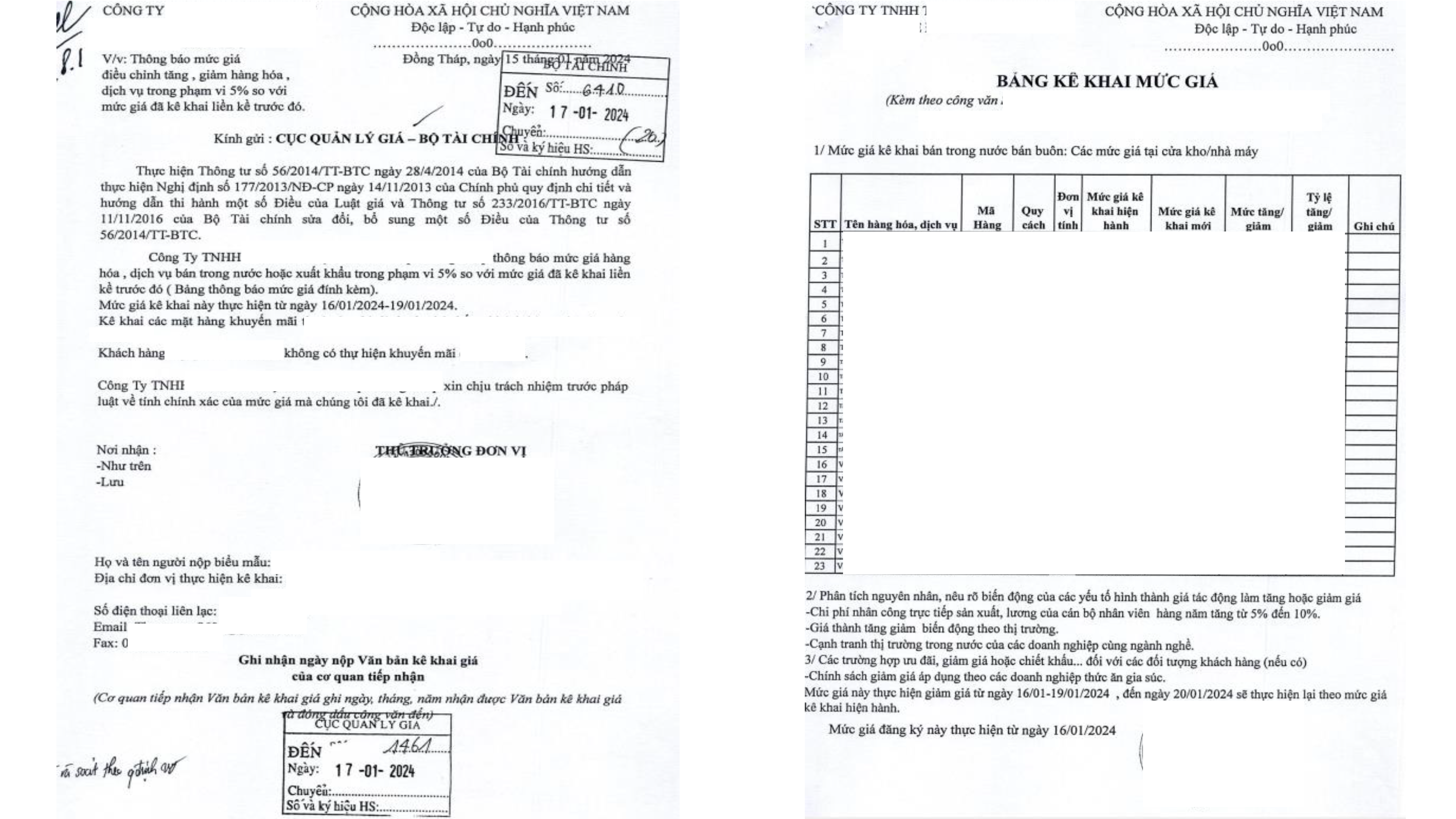
- Required Documents: According to Decree 85/2024/ND-CP.
- Drug Price Declaration Form: As per Decree 85/2024/ND-CP.
Important Notes on Price Declaration Forms
- A single price declaration form can cover multiple products at once.
+
- The selling price of the same product may vary for different buyers.
- Effective Date: From July 10, 2024.
Entities Required for Price Announcement and Declaration
|
Drug Type
|
Wholesale Price Announcement
|
Wholesale/Retail Price Declaration
|
|---|---|---|
|
Prescription drugs in the Essential Medicines List
|
✅ Required
|
✅ Required
|
|
Prescription drugs NOT in the Essential Medicines List
|
✅ Required
|
❌ Not required
|
|
Non-prescription drugs in the Essential Medicines List
|
❌ Not required
|
✅ Required
|
|
Non-prescription drugs NOT in the Essential Medicines List
|
❌ Not required
|
❌ Not required
|
- Exemption from Price Announcement:
- If the drug is imported for non-commercial purposes.
- If the drug is domestically produced and granted an exemption by the government.


Leave A Comment